Introduction
ABO-incompatible kidney transplantation (ABOi KTx) represents a critical advancement in transplant medicine, offering a highly effective strategy to overcome the immunological barrier posed by incompatible blood groups between a patient with end-stage renal disease (ESRD) and a willing living donor. Historically, the presence of pre-formed anti-A or anti-B antibodies in the recipient would trigger hyperacute or accelerated acute humoral rejection, leading to immediate graft loss. Modern desensitization protocols have rendered this barrier manageable.1
By successfully implementing ABOi KTx, the practice significantly expands the pool of available living donors, which is particularly vital in regions facing a pronounced shortage of compatible organs, such as the local population served by Prince Sultan Military Medical City. The acceptance of ABOi KTx as a safe and successful procedure directly addresses the problem of prolonged waiting times for renal grafts, improving patient outcomes and quality of life. The success of these procedures relies heavily on effective pre-transplant depletion of anti-AB antibodies and robust post-transplant immunosuppression.
This prospective observational study was designed to meticulously track the clinical courses and outcomes of nine consecutive patients who received an ABOi KTx at this single center in the Saudi population. The three-year follow-up period allows for a meaningful assessment of both short-term procedural safety and medium-term graft function stability.2
Objectives
The primary objectives of this pilot study were multifaceted, designed to evaluate both the implementation strategy and the clinical efficacy within the local context:
Feasibility Assessment and Safety Validation: To definitively assess the procedural feasibility and clinical safety profile of performing ABO-incompatible transplants at Prince Sultan Military Medical City. This was deemed essential due to the specific challenges and donor dynamics encountered in the region, particularly the ongoing shortage of compatible living donors that necessitated exploring this alternative pathway. Successful implementation here provides a reproducible blueprint for other regional centers.3
Outcome Observation and Comparison: To observe, record, and subsequently compare the incidence rates of acute rejection episodes and the overall medium-term graft survival rates in the ABO-incompatible recipient cohort against the center’s historical benchmarks for ABO-compatible transplants. While direct comparative data is often external, establishing a baseline outcome for the ABOi cohort is crucial for determining if the increased complexity of the procedure translates into acceptable clinical outcomes comparable to standard transplantation. A successful outcome would reinforce the rationale for broader clinical adoption.4
Methods
Study Design
This was rigorously conducted as a single-center, three-year prospective observational study. The cohort included all consecutive ABOi KTx cases performed between 2013 and 2017, ensuring an unbiased selection of patients who subsequently underwent transplant follow-up at the specified medical city.
Desensitization Protocol
The cornerstone of successful ABOi KTx is pre-transplant desensitization aimed at reducing the circulating anti-AB antibody titers below a critical threshold (typically 1:8 or 1:16, but ideally 1:4 or lower). All patients received a mandatory standard basic desensitization protocol, irrespective of their initial antibody levels:
- Rituximab (Anti-CD20 Monoclonal Antibody): Administered at a standard dose (375 mg/m²), although the documented doses predominantly centered around 500mg. Rituximab targets and depletes circulating B-lymphocytes, the precursor cells responsible for producing anti-AB antibodies, thereby preventing antibody rebound after plasma exchange.
- Intravenous Immunoglobulin (IVIG): Administered at 2 g/kg. IVIG saturates Fc receptors on effector cells, modulates T and B cell function, and acts as a passive blockade, effectively neutralizing residual circulating antibodies and providing non-specific immunosuppression.
Titer-Guided Intervention: The decision for more intensive therapy was strictly titer-dependent, utilizing the post-Rituximab/IVIG antibody level:
- Immunoadsorption (IA) or Plasmapheresis (PP): These procedures were initiated for patients whose Anti-AB titer remained above 1:4. This threshold is recognized as a key cut-off for minimizing the risk of antibody-mediated rejection (AMR). IA and PP physically remove circulating antibodies from the plasma. IA, specifically, uses columns to selectively bind and remove immunoglobulins, offering a potentially more targeted and effective approach than non-selective PP. The number of pre- and post-transplant sessions was individualized based on the patient’s continuous titer monitoring and clinical response.
Results
Patient Demographics and Donor Details
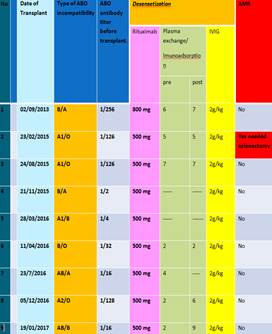
The study cohort comprised 9 patients (5 male, 4 female). The recipients were relatively young, spanning an age range from 14 to 56 years, with a mean age of 33.78 years. The donor pool was exceptionally young, with a mean donor age of only 24.5 years. This young donor age, coupled with the predominance of living donors (8 out of 9), suggests high quality and viability of the transplanted organs. Notably, the inclusion of one deceased donor demonstrates the center’s preparedness to apply the ABOi protocol even in the context of non-living, deceased-donor transplantation where compatibility is often dictated by chance.
Desensitization and Procedures
The necessity for intensive antibody removal procedures correlated directly with the initial anti-AB antibody burden, validating the risk stratification approach:
- High Titer Cohort: 6 out of 9 patients required extensive desensitization, necessitating both pre- and multiple post-transplant IA sessions. These patients likely had high baseline titers, requiring sustained efforts to maintain the antibody levels in the safe zone after transplantation.
- Partial/Medium Titer Cohort: 1 patient required only pre-transplant IA, suggesting the post-operative titer was successfully maintained by basic immunosuppression alone.
- Low Titer Cohort: Crucially, 2 patients (with initial titers of 1/2 and 1/4) required neither pre- nor post-transplant IA. This highlights that low initial titers, which carry a minimal immunological risk, can be successfully managed solely with the basic regimen of Rituximab and IVIG, significantly reducing the procedural burden and cost.
Clinical Outcomes (Graft Function and Rejection)
The overall clinical success rate was high, with the vast majority (8 out of 9) remaining rejection-free during the three-year follow-up.
- Rejection Event: Only one patient (11.1%) experienced acute rejection, specifically Antibody-Mediated Rejection (AMR).
- A1 Immunogenicity: This singular case was highly informative: the kidney was received from a Blood Group A1 donor. The A1 subtype possesses more complex and numerous ABO carbohydrate antigens compared to the A2 subtype, making it significantly more immunogenic. This finding supports the conclusion that A1 donors present a higher immunological challenge in the ABOi setting.
- Rejection Management: The acute rejection in this patient initially proved refractory to conventional rescue therapy, failing to respond even after additional post-transplant Immunoadsorption. The ultimate resolution was achieved only after a splenectomy was performed. The spleen is a major site of B-cell proliferation and antibody synthesis; its removal provided the necessary halt to the pathogenic anti-AB antibody production, successfully reverting (resolving) the acute rejection episode. This demonstrates the critical role of aggressive, secondary interventions for highly immunogenic rejections.
- Long-term Graft Function: At the end of the observation period, the group exhibited excellent mean graft function. The Mean Average Serum Creatinine (sCr) was 93 µmol/L (range 50 – 136 µmol/L). In the context of normal sCr ranges (typically 44–110 µmol/L), a mean of 93 µmol/L is highly favorable, strongly suggesting durable and good long-term function for the transplanted kidneys across the entire cohort.
Conclusions
The observed outcomes strongly support the institutional commitment to this advanced procedure:
- Safety and Feasibility Confirmation: ABO-incompatible kidney transplantation (ABOi KTx) has been definitively established as a safe and fully feasible procedure within the center, successfully expanding the donor pool and serving patients in critical need.
- Rejection Rate Equivalence: The very low acute rejection rate observed (11.1%) is considered similar to, if not slightly better than, typical rates seen in standard ABO-compatible transplants at the center. This underscores that with proper desensitization, the added immunological risk is effectively neutralized.
- Refined Risk Stratification: The study provides two crucial clinical takeaways for procedural refinement:
- Anti-AB Titer is Predictive: A low initial Anti-AB titer (e.g., ≤1:4) significantly correlates with reduced requirement for complex desensitization protocols (IA/PP) and demonstrates a lesser chance of acute rejection. This allows for the selection of lower-risk patients for a less invasive protocol.
- A1 Subtype is the Highest Risk Factor: The fact that the only rejection case stemmed from an A1 donor confirms that Blood Group A1 is the most immunogenic subtype. Future protocols must acknowledge this higher inherent risk, possibly mandating more aggressive initial desensitization and closer post-operative monitoring for A1 donor transplants compared to A2 or B donor transplants. The successful use of splenectomy as a last-resort rescue therapy further emphasizes the severity and unique management challenges associated with this specific incompatibility.4
Authors of this article
AUTHOR NAME-
1.DR. EBADUR RAHMAN, SENIOR CONSULTANT, NEPHROLOGY,EVERCARE HOSPITAL DHAKA,EMAIL-ebadur.rahman@evercarebd.com
CO-AUTHOR-
- DR. RUBAIYA RAHIM
- DR. FAIROOZ RAISA
- Dr. ANZUMAN ARA
- Dr. Ummay Farwa
- Dr. Asma Jalal Panjery
- Dr . A. F M. Abul Foiz MD. Noman
7. DR. REZOYANA NAZIM, SENIOR SPECIALIST, NEPHROLOGY, EVERCARE HOSPITAL DHAKA, EMAIL- dr.esha03@gmail.com
8.DR. AHMED KHALED, SENIOR CONSULTANT, HISTOPATHOLOGY ,EVERCARE HOSPITAL DHAKA,EMAIL-ahmed.khaled@evercarebd.com
9.BRIG. GEN.DR. SM MAHBUBUL ALAM, SENIOR CONSULTANT, HISTOPATHOLOGY, EVERCARE HOSPITAL DHAKA,EMAIL-mahbubul.alam@evercarebd.com
10. Dr. Arif Mahmud
11. Zahid Hossain
12. Dr. Sultana Parveen
References
- Council of Europe – Newsletter Transplant. International Figures on Donation and Transplantation – 2013. Strasbourg: European Directorate for the Quality of Medicines & Health Care, 2014
- Zschiedrich S, Kramer-Zucker A, Janigen B et al. An update on ABO incompatible kidney transplantation. Transpl Int 2015; 28: 387–397
- Tanabe K. Japanese experience of ABO-incompatible living kidney transplantation. Transplantation 2007; 84: S4–S7
- Montgomery RA, Locke JE, King KE et al. ABO incompatible renal transplantation : a paradigm ready for broad implementation. Transplantation 2009; 87: 1246–1255