Bacteria are broadly classified into two groups—Gram-positive and Gram-negative—based on their cell wall structure and staining characteristics. This classification is derived from the Gram staining method developed by Hans Christian Gram in 1884, which remains a cornerstone of microbiological diagnostics today. The technique involves staining organisms with crystal violet or methylene blue dye, allowing visualization under a microscope.
Gram-positive bacteria possess a thick peptidoglycan cell wall, enabling them to retain the blue dye during staining, thus appearing blue or violet under the microscope. They may appear in cocci (spherical), bacilli (rod-shaped), or filamentous forms.
In contrast, Gram-negative bacteria have a thin peptidoglycan layer but an additional outer membrane rich in lipopolysaccharides, which acts as a strong barrier against many antibiotics. Because they cannot retain the primary dye, Gram-negative organisms appear pink or red after Gram staining. The presence of this outer membrane, along with efflux pumps and enzyme-mediated resistance, makes Gram-negative bacteria significantly more difficult to treat and one of the most important public health challenges globally.1
Acinetobacter baumannii: An Emerging Gram-Negative Superbug
Acinetobacter baumannii is a Gram-negative bacterium increasingly recognized as a major cause of severe hospital-acquired infections, particularly in critically ill or immunocompromised patients. Despite being Gram-negative, it can occasionally be misidentified as Gram-positive in laboratory testing. This occurs because A. baumannii sometimes retains the crystal violet stain used in Gram staining—leading to occasional diagnostic confusion.
Acinetobacter species are notable for their environmental resilience. They are found in soil, water, and hospital environments, where they can colonize human skin, medical devices, and environmental surfaces. In healthcare facilities, A. baumannii can survive for weeks to months, even in dry conditions, making it exceptionally difficult to eradicate once established.2
Carbapenem-Resistant Acinetobacter baumannii (CRAB): Why It Matters
When A. baumannii acquires resistance to carbapenems—a potent class of broad-spectrum β-lactam antibiotics such as imipenem, meropenem, and doripenem—it is referred to as Carbapenem-Resistant Acinetobacter baumannii (CRAB).
Carbapenems are widely regarded as the last line of defense against multidrug-resistant bacterial infections because of their ability to inhibit bacterial cell wall synthesis and resist degradation by most β-lactamases. However, the increasing clinical use and misuse of carbapenems have fueled the emergence of resistant strains, including CRAB, which poses a grave public health concern.3
CRAB achieves resistance through several mechanisms, most notably production of carbapenemase enzymes that deactivate carbapenems, and mutations in outer membrane proteins that reduce antibiotic penetration. These adaptations make CRAB infections extremely difficult to treat and control, particularly in intensive care settings where patients often rely on invasive devices such as ventilators or catheters.
In essence, Acinetobacter baumannii becomes a CRAB strain when it develops the ability to withstand carbapenem therapy, transforming a common environmental organism into a multidrug-resistant superbug capable of causing outbreaks in healthcare environments.
Environmental Hardiness and Biofilm Formation
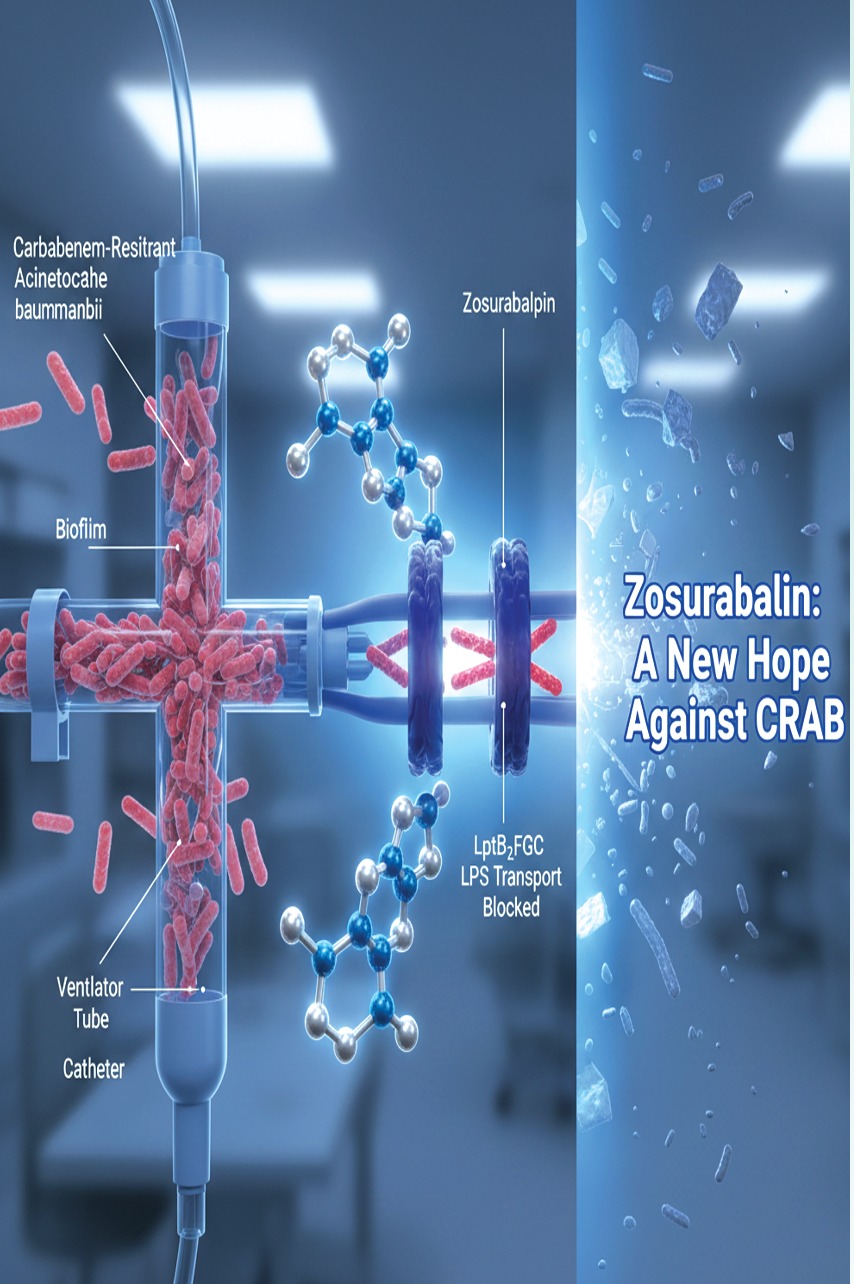
The persistence of Acinetobacter baumannii, particularly in its carbapenem-resistant (CRAB) form, is largely attributed to its exceptional environmental resilience and ability to form biofilms—dense, protective bacterial communities that adhere to medical surfaces such as ventilators, catheters, and intravenous lines. These biofilms shield the bacteria from antibiotics, disinfectants, and host immune responses, enabling long-term colonization and facilitating cross-transmission among patients and healthcare workers.2
A. baumannii can survive for weeks to months on dry surfaces, promoting persistence within healthcare environments. Its ability to colonize high-touch hospital surfaces—including bedrails, monitors, and tables—makes infection control especially challenging.4
Patients at highest risk for CRAB infection include:
- Individuals admitted to intensive care units (ICUs) or with prolonged hospital stays
- Those receiving mechanical ventilation or using urinary catheters and IV lines
- Patients with open wounds, weakened immunity, or chronic illnesses such as diabetes or lung disease.
CRAB can spread through multiple routes within healthcare settings:
- Contaminated hands of healthcare workers
- Direct contact with infected or colonized patients
- Shared medical equipment such as stethoscopes and ventilators
- High-touch surfaces (e.g., bedrails, doorknobs) that remain contaminated for extended periods
This combination of environmental persistence, biofilm formation, and ease of transmission makes A. baumannii one of the most resilient and transmissible pathogens in hospital environments.5
Clinical Manifestations of CRAB Infections
CRAB infections primarily affect critically ill or immunocompromised patients, particularly those in ICUs or postoperative care. The clinical spectrum is broad and often severe, depending on the infection site. Common manifestations include:
- Ventilator-associated pneumonia (VAP)
- Sepsis or bloodstream infections
- Wound and burn infections
- Urinary tract infections (UTIs), especially catheter-associated
These infections are associated with high morbidity and mortality, especially in patients with invasive devices or prolonged hospital stays.6
Due to its extensive multidrug resistance, CRAB leaves limited therapeutic options available. Treatment often requires combination antibiotic regimens, use of new or investigational agents, and strict infection prevention protocols to minimize transmission risk.
From CRAB to Zosurabalpin: A New Hope in a Dire Landscape
Given the formidable challenge posed by carbapenem-resistant A. baumannii (CRAB)—its ability to persist in hospitals, evade multiple antibiotic classes, and cause life-threatening infections—the emergence of Zosurabalpin, a novel antibiotic under trail almost after 50 years of the last antibiotic discovered, represents a compelling and much-needed breakthrough.
Class and Design
Zosurabalpin is a tethered macrocyclic peptide (MCP)—a small cyclic peptide engineered to be zwitterionic (bearing both positive and negative charges) and plasma-compatible. The tether (linker) locks the macrocycle into a conformation favorable for binding its bacterial target. Through chemical optimization from initial screening hits, Roche and collaborators advanced to second-generation zwitterionic MCPs, of which zosurabalpin is a leading candidate.7
Mode of Action
Zosurabalpin exerts its bactericidal effect by targeting a novel and previously unexploited vulnerability in Gram-negative bacteria: the LptB₂FGC lipopolysaccharide (LPS) transporter complex, an ATP-dependent inner-membrane machine responsible for extracting LPS from the inner membrane and shuttling it across the periplasm to assemble the outer membrane. By trapping LPS within the transporter and blocking its transit, zosurabalpin disrupts the formation and maintenance of the bacterial outer membrane—rendering it lethal to the cell. This mechanism is unlike that of conventional antibiotics and avoids many existing resistance pathways.7
In structural studies, the drug was shown to make extensive contacts with both Lpt components (LptF/G) and the bound LPS, confirming its dual binding mode.7
Current Clinical Status and Use
Zosurabalpin is still investigational and not approved for routine use. Roche is preparing to initiate a Phase 3 trial, planning to enroll approximately 400 patients with invasive CRAB infections in hospital settings. The trial will compare zosurabalpin versus standard-of-care therapies.8
Its principal indication is intended to be severe hospital-acquired CRAB infections, such as ventilator-associated pneumonia, bloodstream (septic) infections, and other nosocomial A. baumannii disease contexts where existing antibiotics are ineffective. The WHO ranks CRAB as a priority pathogen, and Roche’s development plans reflect that urgency.9
In trials, zosurabalpin is administered intravenously (IV)—typically over a one-hour infusion, with dose escalation studied across patients. No oral formulation is available (and would likely be challenging, given the macrocyclic peptide nature).10
Stewardship and Use Strategy
Because zosurabalpin has a narrow spectrum—primarily targeting A. baumannii—its clinical deployment is expected to be strategic and conservative. The most plausible model is empirical use only in very high-risk, suspected CRAB cases, or targeted therapy after culture/susceptibility confirmation. This approach aligns with antibiotic stewardship goals of preserving novel agents and limiting resistance pressure.10
Kinetics, Pharmacokinetics, and Safety
Kinetics, Pharmacokinetics, and Safety (Simplified)
- How fast it works:
Lab studies showed that zosurabalpin kills bacteria quickly, with the number of bacteria dropping within a few hours at effective doses. In infected mice, treatment reduced bacterial counts within 24 hours and helped them survive.11 - How the body handles it (PK / Half-life):
Early studies in animals and humans found that zosurabalpin leaves the body fairly quickly. It reaches its highest blood level at the end of the 1-hour IV infusion, and then levels fall fast. The drug showed high clearance (the body removes it quickly) and a short half-life (around 0.3 hours in animals). Results were similar in people, though there were some differences between individuals. Mild liver problems did not require dose changes in small studies.11 - Safety so far:
Early human trials found zosurabalpin was well tolerated, even at high doses (up to 2,000 mg). The most common side effects were mild infusion reactions, and no serious drug-related problems were reported. Detailed safety information will come after Phase 3 trials and full regulatory review.12
Summary of Zosurabalpin
Zosurabalpin is a first-in-class, tethered macrocyclic peptide antibiotic that inhibits the LptB₂FGC LPS transporter, producing potent activity—both in vitro and in vivo—against carbapenem-resistant A. baumannii. With its unique mechanism, narrow spectrum, and early favorable safety/PK profile, it holds promise as a new weapon against CRAB. However, its ultimate success depends on Phase 3 outcomes, stewardship policies, and commercial viability.
Conclusion: The Stakes, the Barriers, and the Promise
The rise of antimicrobial resistance (AMR)—particularly among Gram-negative pathogens—represents one of the gravest threats to modern medicine. Acinetobacter baumannii, especially in its CRAB form, epitomizes this crisis: it resists numerous antibiotic classes, thrives in healthcare environments, and causes devastating infections such as pneumonia, sepsis, and device-associated disease.
Yet, development of novel antibiotics has lagged for decades, especially for Gram-negative organisms. The physiological barriers (outer membrane, efflux pumps, low permeability), high cost, regulatory risk, and limited commercial incentives have discouraged many pharmaceutical efforts. Consequently, new agents addressing core resistance mechanisms are rare.
In this bleak landscape, zosurabalpin offers a glimmer of hope. Its novel mode of action, targeting LPS transport rather than conventional cell wall synthesis or protein machinery, places it outside the reach of many pre-existing resistance mechanisms. Early data suggest it can overcome even pan-resistant CRAB isolates. If successful, it may become the first new class active against Gram-negatives in decades, and catalyze renewed momentum in antibiotic discovery.
But caution is warranted. For zosurabalpin to live up to its promise, it must prove efficacy and safety in large clinical trials, maintain an acceptable resistance risk, and be integrated into care under robust stewardship frameworks. Moreover, economic and regulatory structures must evolve to support sustainable antibiotic development and access.
In sum: A. baumannii and similar superbugs are a formidable adversary—yet with targeted innovations like zosurabalpin, we may finally gain ground. The battle against AMR is far from over, but each new weapon matters.
Author of this article
Dr. Maliha Mannan Ahmed
MBBS (BMC), MBA (ULAB), Masters in Healthcare Leadership (Brown University, USA) and Level 1 Certification on Precision Nutrition.
The Executive Editor of The Coronal.
Reference
1. Gram-positive and gram-negative: What is the difference? Accessed October 7, 2025. https://www.medicalnewstoday.com/articles/gram-positive-vs-gram-negative
2. Carbapenem-Resistant Acinetobacter baumannii | Wisconsin Department of Health Services. Accessed October 7, 2025. https://www.dhs.wisconsin.gov/disease/carbapenem-resistant-acinetobacter-baumannii.htm
3. Thacharodi A, Vithlani A, Hassan S, Alqahtani A, Pugazhendhi A. Carbapenem-resistant Acinetobacter baumannii raises global alarm for new antibiotic regimens. iScience. 2024;27(12):111367. doi:10.1016/J.ISCI.2024.111367
4. Carbapenem-resistant Acinetobacter baumannii (CRAB): An urgent public health threat in United States healthcare facilities | A.R. & Patient Safety Portal. Accessed October 7, 2025. https://arpsp.cdc.gov/story/cra-urgent-public-health-threat
5. Ngiam JN, Chung M, Koh Y, Chew KL, Yi MC. Clinical characteristics and treatment outcomes of Acinetobacter baumannii bloodstream infections in a setting with high carbapenem susceptibility among isolates. Liu PY, ed. Microbiol Spectr. Published online September 29, 2025. doi:10.1128/SPECTRUM.01607-25
6. Lodise TP, Nguyen ST, Margiotta C, Cai B. Clinical burden of Acinetobacter baumannii, including carbapenem-resistant A. baumannii, in hospitalized adult patients in the USA between 2018 and 2022. BMC Infect Dis. 2025;25(1). doi:10.1186/S12879-025-10749-1
7. Zampaloni C, Mattei P, Bleicher K, et al. A novel antibiotic class targeting the lipopolysaccharide transporter. Nature. 2024;625(7995):566-571. doi:10.1038/S41586-023-06873-0;TECHMETA
8. Roche to launch phase 3 trial for new antibiotic targeting Acinetobacter baumannii | CIDRAP. Accessed October 7, 2025. https://www.cidrap.umn.edu/antimicrobial-stewardship/roche-launch-phase-3-trial-new-antibiotic-targeting-acinetobacter
9. Fierce Pharma Biopharma News & Insights. Accessed October 7, 2025. https://www.fiercepharma.com/
10. Roche takes new antibiotic into phase 3 for “urgent threat” | pharmaphorum. Accessed October 7, 2025. https://pharmaphorum.com/news/roche-takes-new-antibiotic-phase-3-urgent-threat
11. Zampaloni C, Mattei P, Bleicher K, et al. A novel antibiotic class targeting the lipopolysaccharide transporter. Nature. 2024;625(7995):566-571. doi:10.1038/S41586-023-06873-0;TECHMETA
12. Roche designs new antibiotic can fight drug-resistant infections. Accessed October 7, 2025. https://www.fiercebiotech.com/research/roche-designs-new-antibiotic-fight-deadly-baumannii-infections